CLEVELAND, April 03, 2025 (GLOBE NEWSWIRE) -- The hair loss industry is at a tipping point. A recent Wall Street Journal exposé has reignited concerns about the severe and long-term side effects of finasteride, the FDA-approved drug for male pattern baldness. Despite 20 years of documented risks - from hormonal imbalances to mental health issues, it remains the industry standard.
Now, a groundbreaking clinical trial is proving that an effective, drug-free alternative is not only possible but already here. HairSmart is conducting these groundbreaking clinical trials at three IRB board approved clinical sites.
Preliminary results from a six-month study involving 72 patients across three clinical sites, led by top hair transplant surgeons, reveal that a 100% plant-based DHT blocker can significantly reduce hair loss and stimulate new growth—without pharmaceutical risks.
Early three-month data from seven patients shows an unprecedented 26% increase in new hair count without finasteride or minoxidil.
A Paradigm Shift in Hair Loss Treatment
“This trial is about more than just hair loss—it’s about medical ethics, patient safety, and scientific progress,” says Prerna Khemka, CEO and Founder of HairSmart.
“For decades, safer alternatives to finasteride have been ignored despite mounting risks. The recent WSJ article is just the latest warning! Telehealth companies continue to push these drugs while downplaying side effects. The real question is: why have only two FDA-approved drugs existed for hair loss in over 30 years? It’s time for change.”
Trial Breakthroughs: First-of-Its-Kind Plant-Based Approach
This study is setting industry milestones:
- First clinical trial rigorously testing a plant-based DHT blocker for regrowth.
- First designed as a finasteride-alternative protocol.
- One of the largest studies of its kind, tracking 72 patients over six months.
- First clinical proof that genetic hair loss can be reversed without pharmaceuticals aka finasteride and minoxidil.
Study Design & Key Results
The male protocol includes a plant-based DHT blocker and Ayurvedic hair growth formula, while the female protocol includes these plus a topical growth serum.
Early Results (First 3 Months):

Dr. Ronald Shapiro, MD, lead investigator, states: “A 26% increase in new hair growth in three months using botanicals is impressive. If results continue, we will witness a true paradigm shift.”
Case Study Highlights:
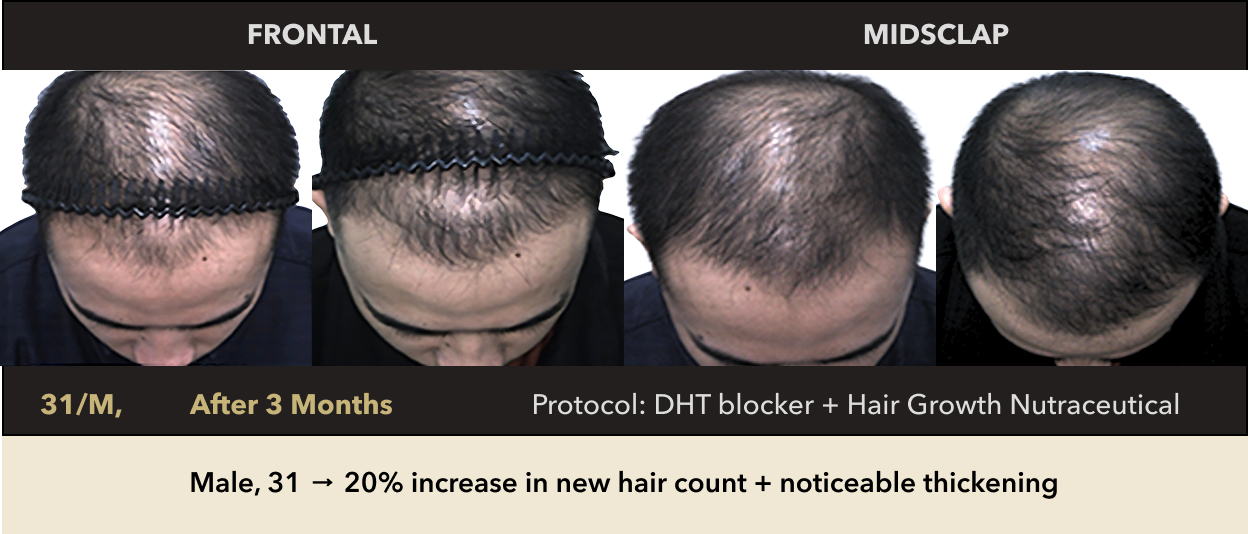
Male, 31 → 20% increase in new hair count + noticeable thickening
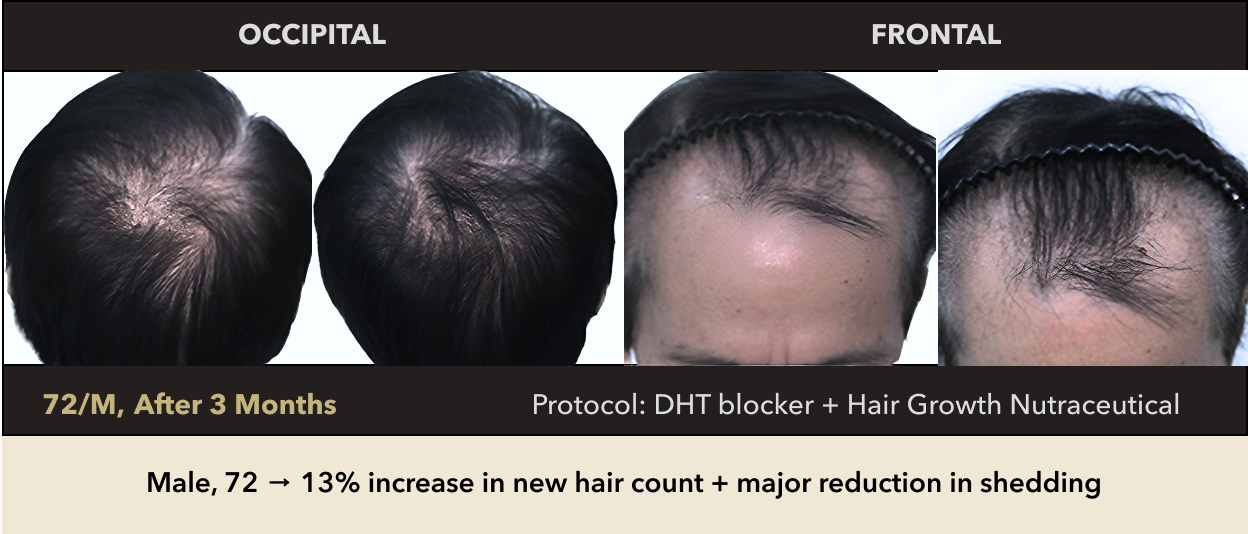
Male, 72 → 13% increase in new hair count + major reduction in shedding
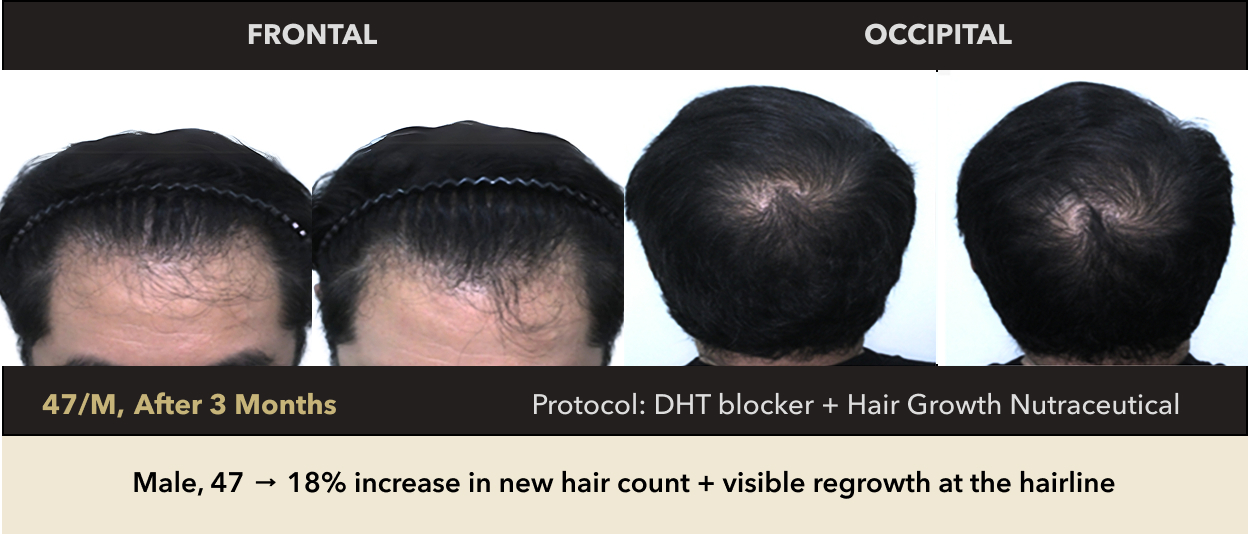
Male, 47 → 18% increase in new hair count + visible regrowth at the hairline
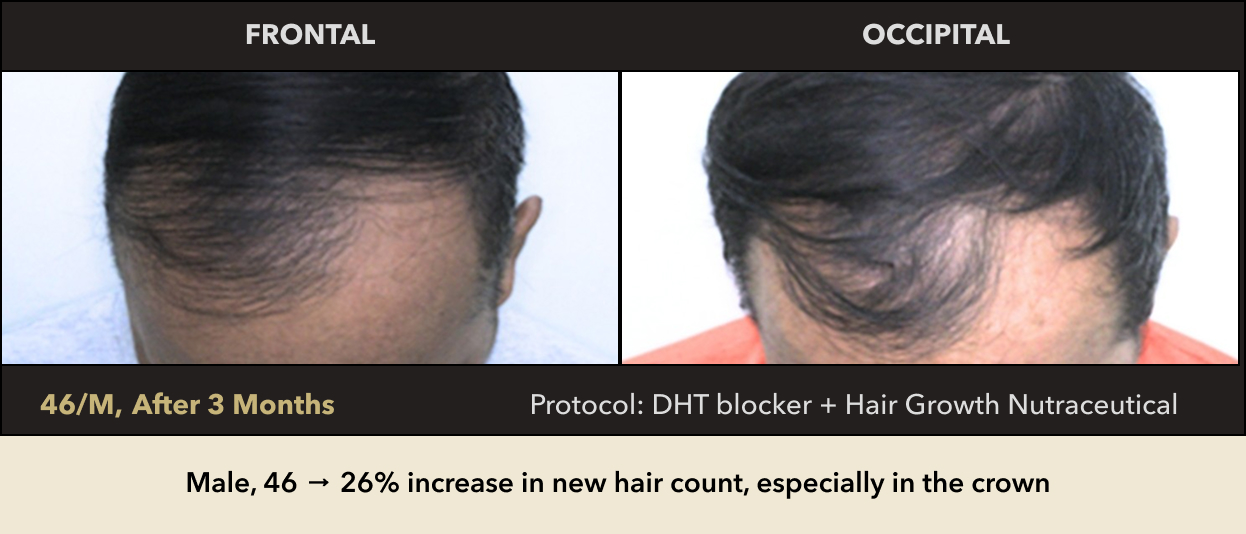
Male, 46 → 26% increase in new hair count, especially in the crown
Female, 31 → 15% increase in new hair count with a confidence boost

Female, 50 → 14% increase in new hair count, with a confidence boost

Female, 67 → 6% increase in new hair count with zero side effects


Zinaria Williams, MD, Hair Transplant & Oculofacial Plastic Surgeon and Clinical Herbalist, echoes this sentiment, underscoring the importance of plant-based innovation in medicine: "To be on the cutting edge of health and medicine, we must be open to plant-based alternatives as a course of action. As a hair transplant surgeon and clinical herbalist, I examined the HairSmart ingredients and immediately knew they were on to something. They are taking a holistic approach to hair growth, and my patients have been getting excellent results. I am happy but not surprised by this preliminary data. I look forward to the full study and congratulate them on being pioneers in this movement."
A Turning Point for the Hair Loss Industry
Unlike traditional drugs, this plant-based approach is proving effective without hormonal risks.

Dr. Mariel Arambulo, lead investigator, states: “The results with this natural protocol rivals traditional drug-based treatments. This is a major leap forward.”
Dr. Piyush Kumar, a top hair transplant specialist, agrees: “For years, doctors and patients have sought a safe finasteride alternative. This study is promising and early data suggests that a plant-based solution works.”
What’s Next? A New Era of Drug-Free Hair Restoration
With three months of data exceeding expectations, the six-month study continues, making it the largest clinical trial of its kind.
The hair loss industry is projected to reach $12.99 billion by 2028. These findings could shift consumers away from pharmaceuticals toward natural solutions.
Prerna Khemka’s vision: “Hair loss isn’t just about vanity—it’s deeply personal. Only two FDA-approved drugs in 30+ years is shocking and unacceptable. Plant-based alternatives deserve the same rigorous trials and FDA consideration as pharmaceuticals.
To achieve this, mission-driven companies need funding and resources equal to pharmaceutical giants. Hair loss treatment shouldn’t be limited to outdated drugs with known risks - it’s time for a safer, science-backed, plant-based revolution.
Our goal is simple: empower people to take control of their hair health - naturally, safely, and effectively. And now, we have the clinical evidence to prove it.”
Stay Tuned: Full Six-Month Results Coming Soon
The final results could reshape hair loss treatments forever.
About Hair Smart: Hair Smart is a pioneering biotech company developing clinically proven, plant-based solutions for hair regrowth. By combining cutting-edge research with natural ingredients, Hair Smart is revolutionizing the industry without pharmaceuticals.
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/4d6d2447-6d0b-4c3c-bd5e-01d042a00dd2
https://www.globenewswire.com/NewsRoom/AttachmentNg/e431be1c-1400-4999-ac28-c48fd76bb160
https://www.globenewswire.com/NewsRoom/AttachmentNg/5499802d-2153-448a-8557-a88424309012
https://www.globenewswire.com/NewsRoom/AttachmentNg/2e3b697f-52f3-4c5c-9784-2e18494c1c28
https://www.globenewswire.com/NewsRoom/AttachmentNg/67b851a5-ffb9-4ffb-99d8-b546375e8f80
https://www.globenewswire.com/NewsRoom/AttachmentNg/e71594b3-1526-4afc-b135-5b9745d3c43f
https://www.globenewswire.com/NewsRoom/AttachmentNg/b7dff24d-3632-48d2-aa72-68d1a0754e1f
https://www.globenewswire.com/NewsRoom/AttachmentNg/470a7195-b805-401b-88c1-71c1068b3498
https://www.globenewswire.com/NewsRoom/AttachmentNg/70615f3c-6e10-4418-8e95-ee305d1d5736
https://www.globenewswire.com/NewsRoom/AttachmentNg/0af11a71-cd0d-403c-8ef3-7c94d80c6161
https://www.globenewswire.com/NewsRoom/AttachmentNg/c861adaa-c229-456f-98ec-8dc5cfc25f06
https://www.globenewswire.com/NewsRoom/AttachmentNg/35d72e7f-589b-4a8b-8f6a-c4e70b14b20a
A video accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/7d6d7dc0-784e-45d0-8d26-5f6eb2057b0c

For Media, PR & Investing Opportunities: contact manishg@myhairsmart.com