Neuronoff Demonstrates Multi-Site IPG Compatibility for Injectrode® Platform, Expanding Neuromodulation Beyond Spinal Cord Stimulation
PR Newswire
CLEVELAND, March 16, 2026
Needle-Delivered Tripolar Electrode Connects to Standard IPG Connectors; Selectively Stimulates Lumbar DRG, Peripheral Nerve, and Sacral Targets from Single Implantable Pulse Generator
CLEVELAND, March 16, 2026 /PRNewswire/ -- Neuronoff, Inc., a clinical-stage neuromodulation company, today announced successful chronic large animal demonstration of its IPG-compatible Injectrode® platform, in which a single remotely controlled Implantable Pulse Generator (IPG) independently stimulated three anatomically distinct neural targets via standard Bal Seal connectors.
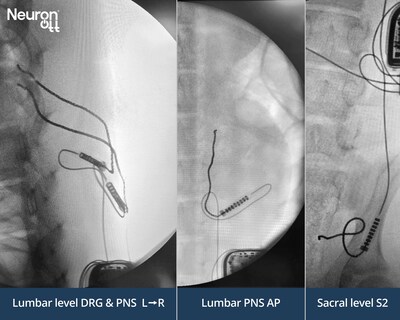
Multi-contact Injectrodes were placed at the Lumbar Dorsal Root Ganglion (DRG), the Lumbar Dorsal Ramus, and the Sacral S2 foramen — sites routinely accessed by interventional pain physicians today using needle-based diagnostic blocks, or traditional neuromodulation implants requiring surgery to implant. Each target was stimulated selectively and independently from a single IPG, demonstrating the platform's current steering and multi-channel capabilities.
Expanding the IPG Addressable Market Beyond the Spine
The significance of this demonstration is not limited to the Injectrode platform itself. Because the IPG-Injectrode is delivered entirely through a ~1.5mm needle and connects to IPGs via industry-standard Bal Seal connectors, any IPG manufacturer gains the ability to address peripheral nerve stimulation (PNS) targets as well as the DRG, sacral nerves, lumbar dorsal rami, ganglia on the sympathetic chain, and beyond — that have historically required open surgical approaches or remained outside the reach of implantable systems in clinical practice entirely.
"Spinal cord stimulation (SCS) has been the primary market for IPGs for decades, but the vast majority of treatable nerve targets with easy access are peripheral," said Manfred Franke, CEO of Neuronoff. "The IPG-Injectrode removes the surgical barrier in PNS, enables novel multi-contact approaches for DRG and SCS, and expands the reach of any IPG platform into indications that weren't previously accessible. Any IPG can now reach any nerve a physician can access with a needle — and that is a fundamentally different addressable market."
Reimbursement Infrastructure Already in Place
The Injectrode platform is designed to align with established CPT reimbursement codes 64555 and 64590, covering peripheral nerve stimulation trial, treatment and implant procedures. This existing code coverage removes a critical barrier that has historically slowed adoption of novel neuromodulation approaches.
Path to Commercialization
Neuronoff's near-term focus remains the 510(k) submission of its externally powered Injectrode PNS system, targeting FDA submission in mid 2026. The IPG-compatible platform — designed for patients who progress to fully implanted, continuous stimulation — is positioned as the next stage of the same patient journey, enabling a patient to experience the impact of neuromodulation years earlier without a surgical intervention, and the option to convert easily to a battery-powered chronic option thereafter.
"Our external EPG system and IPG-compatible system are designed as a continuum," Franke added. "The patient journey begins with a minimally invasive step not unlike a steroid injection or needle-based RF ablation — no suturing, no scarring, fully compatible with clinic workflows. Conversion to IPG-based chronic therapy is a natural next step, and we've built the platform so that step requires no changes to existing IPG hardware and no new reimbursement codes, and significantly reduces procedural time compared to current surgical approaches."
About Neuronoff, Inc.
Neuronoff, Inc. (Cleveland, Ohio, USA) is developing the Injectrode®, a minimally invasive peripheral nerve stimulation platform that delivers neuromodulation therapy through a standard clinical needle — without surgery, without scarring, and without opioids. The Injectrode's helical platinum-iridium architecture physically anchors to tissue, addressing the lead migration that accounts for a significant share of neuromodulation device recalls industry-wide.
The platform spans diagnostic trial through chronic implant and IPG-connected therapy across pain, autonomic, bladder, cardiac, and inflammatory indications — all accessible via needle delivery, aligned with existing CPT codes, and built on an IPG-agnostic architecture compatible with standard connectors across the existing IPG ecosystem.
The Injectrode is limited by Federal (US) law to investigational use only. These statements have not been evaluated by the FDA.
Contact: Manfred Franke | manfred@neuronoff.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/neuronoff-demonstrates-multi-site-ipg-compatibility-for-injectrode-platform-expanding-neuromodulation-beyond-spinal-cord-stimulation-302714097.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/neuronoff-demonstrates-multi-site-ipg-compatibility-for-injectrode-platform-expanding-neuromodulation-beyond-spinal-cord-stimulation-302714097.html
SOURCE Neuronoff